TREATMENT flexibility
MagVenture TMS
machines allow
depression treatment
in days, not weeks
FDA-cleared accelerated TMS for Major Depressive Disorder (MDD),
delivered over a flexible treatment schedule, as fast as 5 days.

TREATMENT flexibility
MagVenture TMS
machines allow
depression treatment
in days, not weeks
FDA-cleared accelerated TMS for Major Depressive Disorder (MDD),
delivered over a flexible treatment schedule, as fast as 5 days.
GROUNDED IN RESEARCH, COMMITTED TO TREATMENT, FOCUSED ON RESULTS
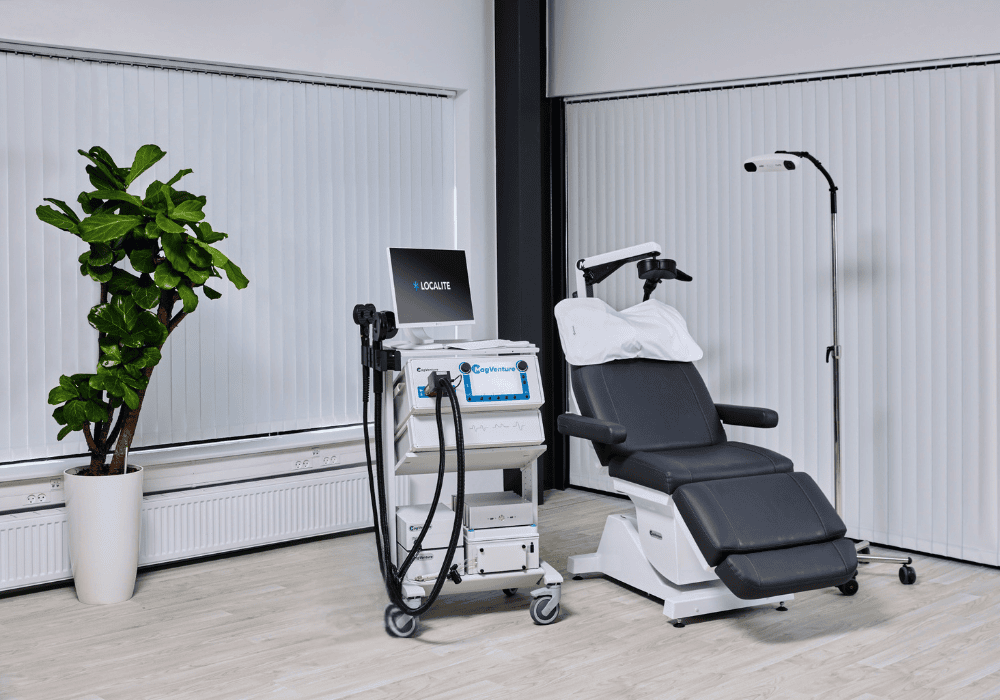
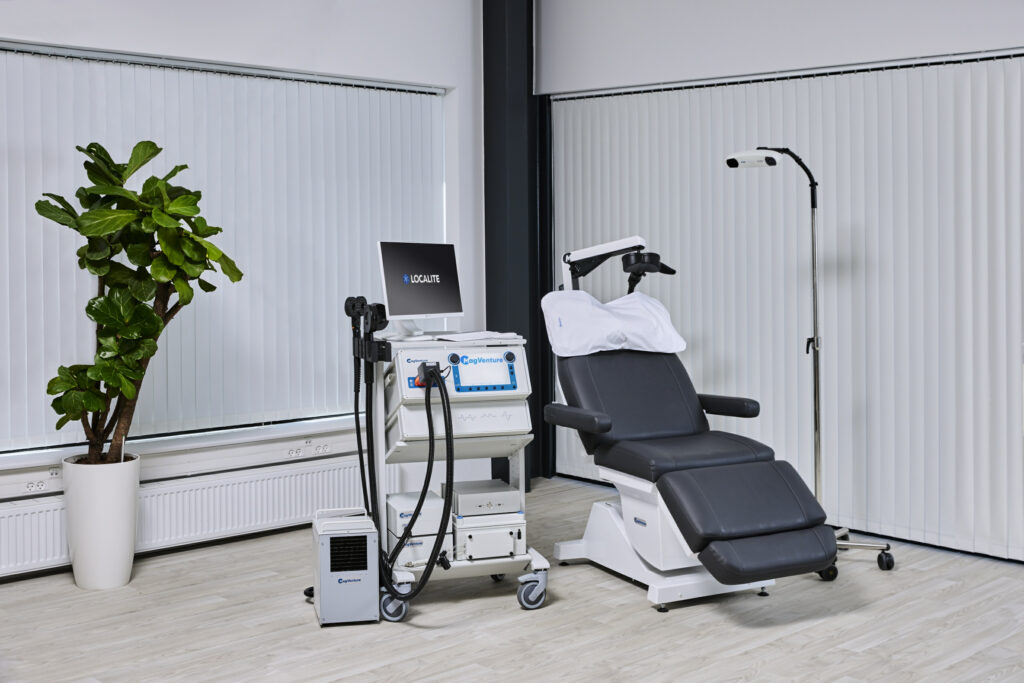
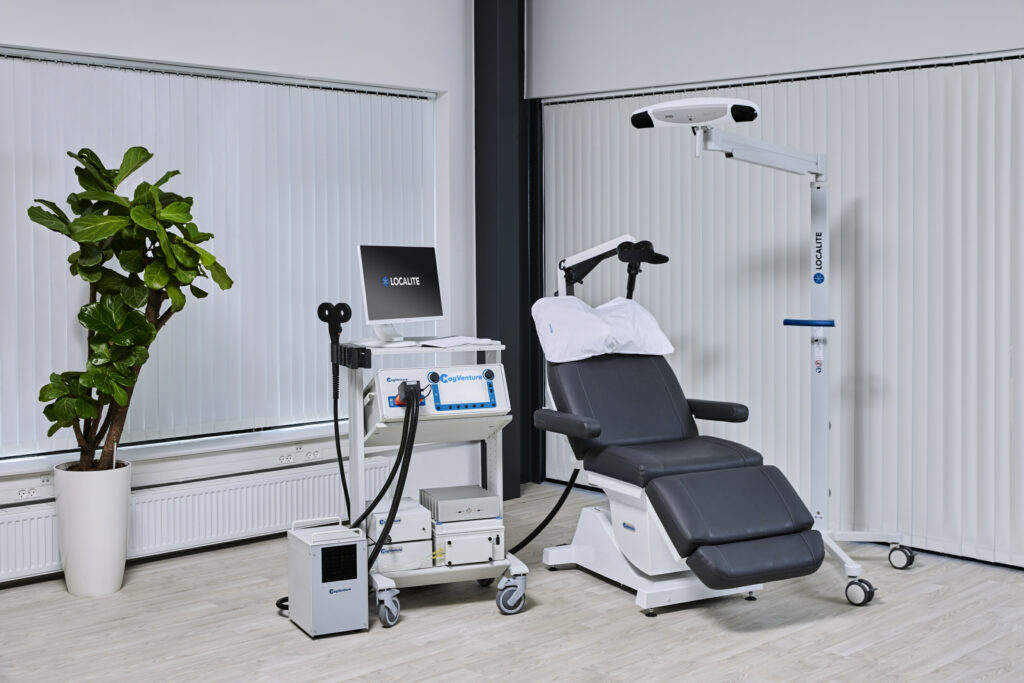
MagVenture is a TMS machine manufacturer.
At MagVenture we are passionate about groundbreaking technology that helps improve people’s lives. That’s why we’ve been pioneering non-invasive Transcranial Magnetic Stimulation (TMS) technology for more than 30 years.
Since the earliest days of TMS, we’ve worked with thousands of researchers to expand their knowledge of the human brain. We have collaborated with leading neuroscientists to advance the fields of psychiatry, neurophysiology, neurology, neuroplasticity, cognitive neuroscience, and rehabilitation.
After many years grounded in the TMS field, we applied our expertise from research and diagnostics to develop clinical TMS systems. More recently we have added Magnetic Peripheral Nerve Stimulation (mPNS) solutions as well.
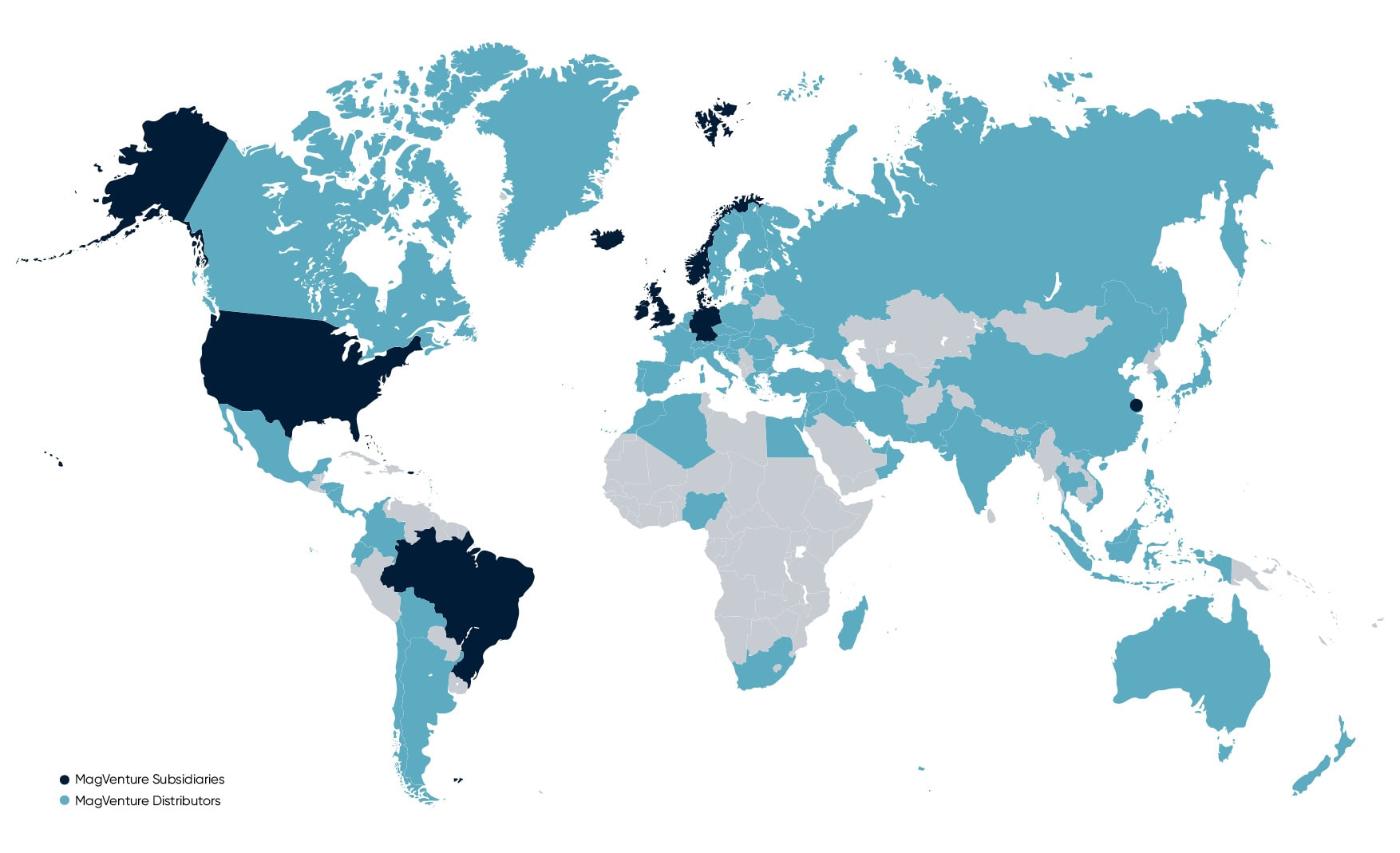
MagVenture is proud to be a privately-owned Danish company. Our products are designed, developed, produced, and quality tested in Denmark to the highest standards. And with subsidiaries in the USA, Brazil, the UK, China, and Germany, and a distribution network in more than 70 countries – our reach is global.

MagVenture Receives FDA Clearance for Accelerated TMS Protocols, Expanding Flexibility for Clinics and Patients
U.S. Food and Drug Administration (FDA) has granted clearance for an expanded indication of its MagVenture TMS Therapy® system to include accelerated transcranial magnetic stimulation (aTMS) protocols for the treatment of Major Depressive Disorder (MDD) in adult patients.
This clearance introduces a more flexible treatment paradigm—enabling clinicians to deliver multiple TMS sessions per day over a condensed schedule. Accelerated protocols, in which multiple TMS sessions (≥2) are administered per day, allow treatment to be completed in as few as 5 days, no MRI or neuronavigation required.

MagVenture receives FDA clearance to expand TMS Therapy indication for Adolescents aged 15–21
The MagVenture TMS Therapy™ system is now indicated as an adjunct treatment for Major Depressive Disorder (MDD) in adolescent patients aged 15 to 21 who have not responded adequately to prior antidepressant medication.
This clearance will provide effective and non-pharmacological therapy to adolescent patients, supporting a growing clinical demand for alternative depression treatments in younger populations.

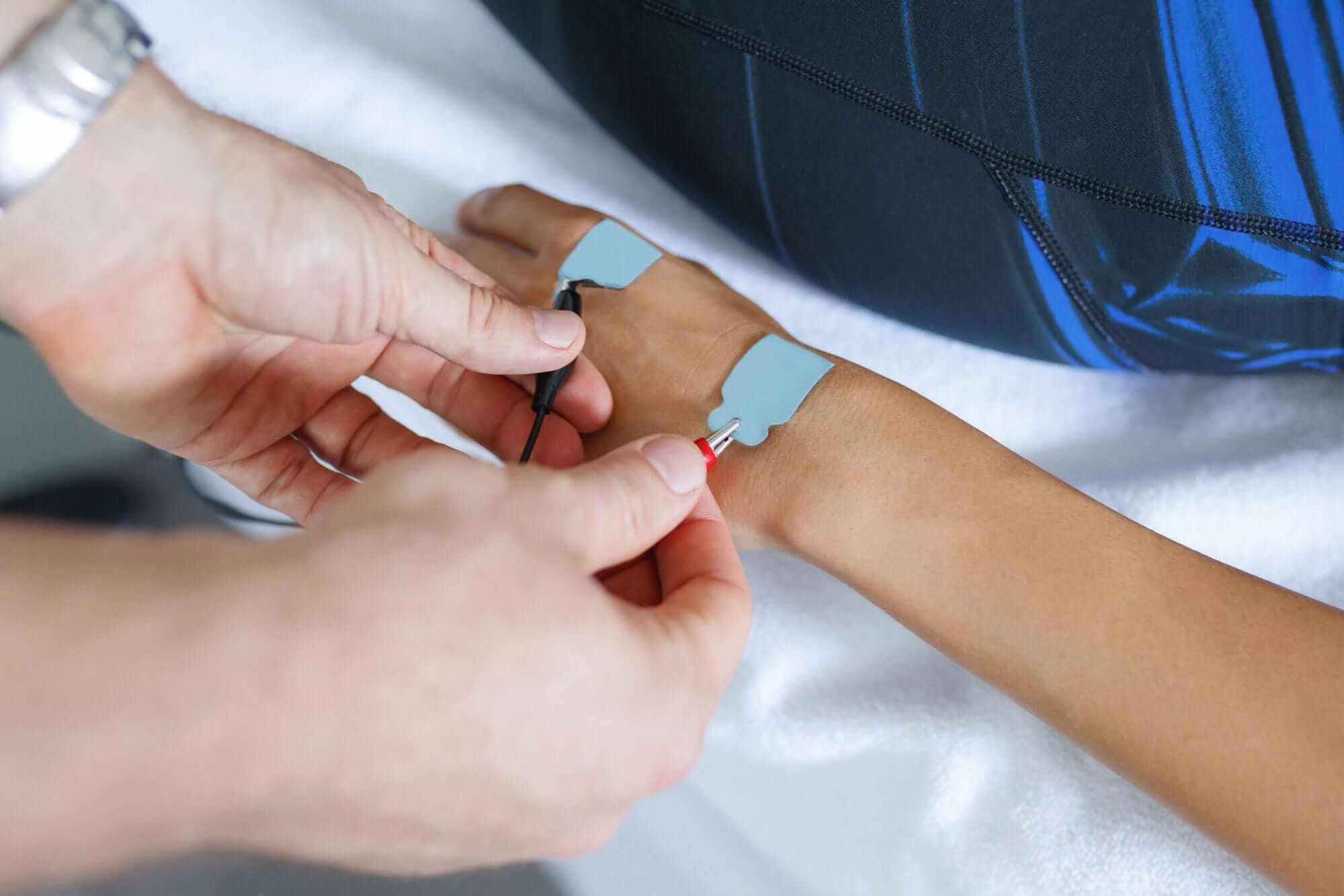
New T65 Coil with Integrated MT Determination Function
MagVenture announces the T65 Coil, the first release in the company’s upcoming T-series—a new generation of lighter, high-performance cooled coils.
Engineered with ergonomic design and active cooling, the T65 combines the features of both the C-B60 and Cool-B65 coils, making it suitable for both motor threshold measurements and treatment. It delivers approximately 50% more pulses than the Cool-B65 and it is developed to support operator comfort and efficient workflow.